Healthcare Leaders
Learn how PIE can help prevent failed colonoscopy preps and reduce hospitalization time.
In-Patient Uses of PIE
1) Colonoscopy Preparation
In-patient colonoscopy prep is both undesirable for the patient and slow for the hospital. With PIE, patients can be prepared for colonoscopy in under 30 minutes.
2) Fecal Impaction Treatment
Severe constipation and fecal impaction often delay discharge for patients. When suppositories and enemas fail, hospitals and spinal cord centers should consider PIE.
Colonoscopy Preparation
Multiple clinical trails have shown PIE to be safe, effective, and with minimal complications for colonoscopy preparation.
Mechanical colon cleansing for screening colonoscopy: a randomized controlled trial
Date Published: December 18 2017
Principle Investigator: David Y. Graham – Baylor College of Medicine and Michael E. DeBakey VA Medical Center
Objective: Compare pulsed irrigation evacuation (PIE), polyethylene glycol (PEG) and sodium phosphate (NaP) colon preparations.
Participants: 391 patients participated in the study (129 in the PEG group, 127 in the sodium phosphate and 135 in the PIE group)
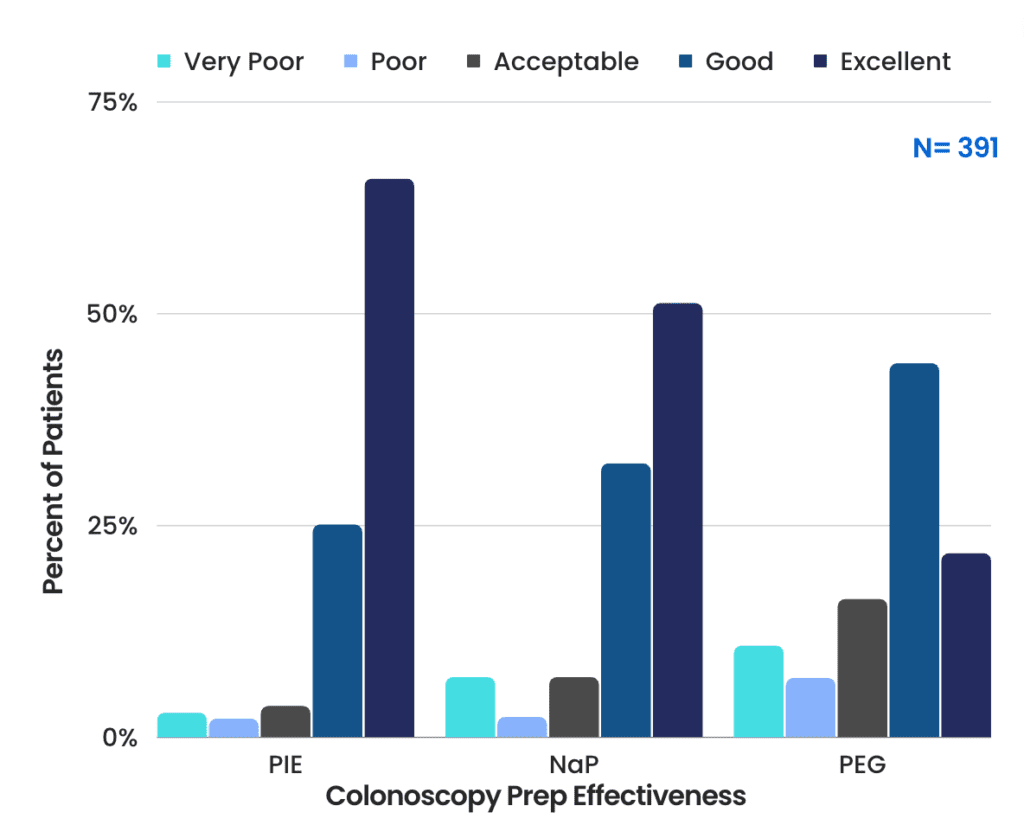
Effectiveness of Prep
- Good or Excellent Prep
- PIE: 91.0%
- NaP: 83.5%
- PEG: 65.8%
- Poor or Very Poor Prep
- PIE: 5.3%
- NaP: 9.4%
- PEG: 16.9%

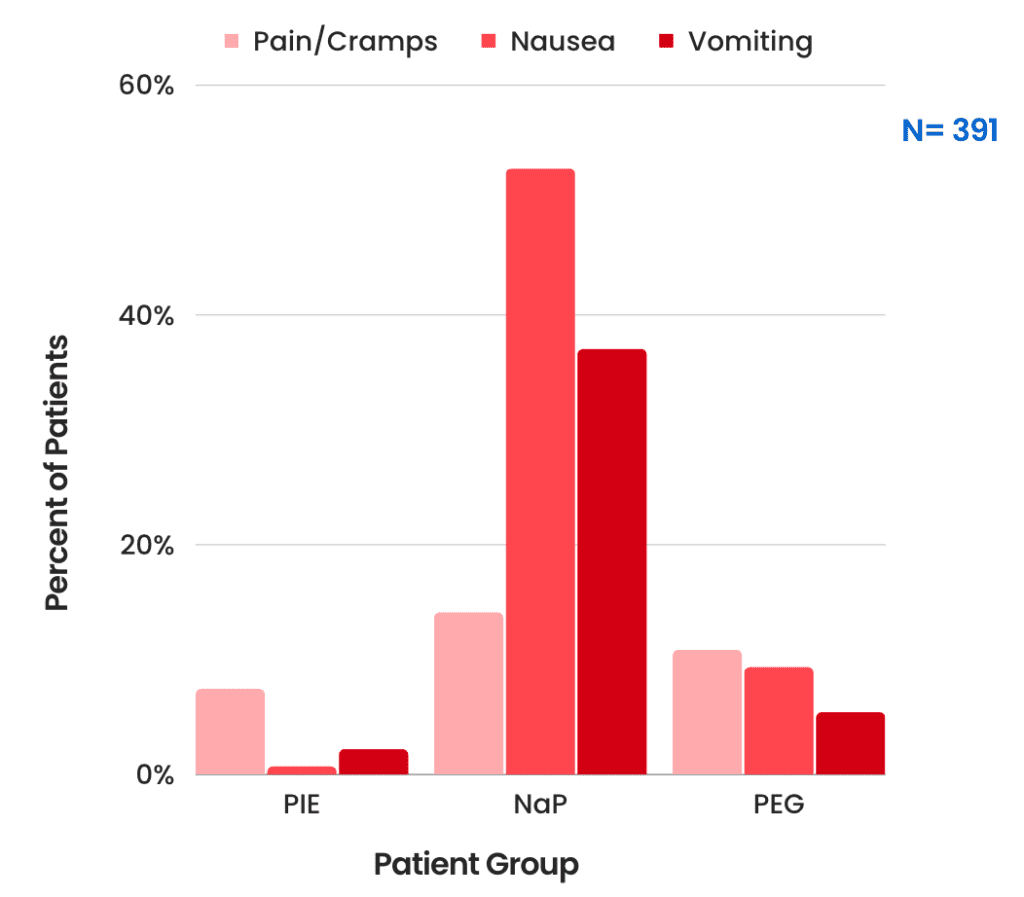
Side Effect Comparison
- Pain/Cramps:
- PIE: 7.4%
- NaP: 14.1%
- PEG: 10.8%
- Nausea
- PIE: 0.7%
- NaP: 52.7%
- PEG: 9.3%
- Vomiting
- PIE: 2.2%
- NaP: 37.0%
- PEG: 5.4%

Email us at info@piemed.com to learn how you can get a PIE Device for your organization.
Citation: Ayub K, Ketwaroo G, Abudayyeh S, Ibrahim A, Cole RA, Brumfield-Brown R, Qureshi WA, Rabeneck L, Graham DY. Mechanical colon cleansing for screening colonoscopy: A randomized controlled trial. J Dig Dis. 2017 Dec;18(12):691-697. doi: 10.1111/1751-2980.12562. PMID: 29160622.
Fecal Impaction Treatment
The PIE Device has been shown to effectively treat acute and chronic fecal impaction through multiple clinical studies.
We wanted to answer 3 questions with our research studies.
1) Who is it for?
2) Is it safe?
The longest PIE study looked at biopsies of the colon after an average of 6.7 years of using the PIE Device. No microscopic, macroscopic, or metabolic abnormalities were noted.
3) Does it work?
Long-term safety of pulsed irrigation evacuation (PIE) use with chronic bowel conditions
Background and Methodology: In this study, pulsed irrigation evacuation (PIE) was used by patients with spinal cord injuries to assess long-term safety. The PIE procedure was performed an average of 3.5 times per week for an average of 6.7 years. To assess microscopic damage, patients received a colonic biopsy.
Results: Pathology results for all of the patients showed no evidence of epithelial damage or abnormal inflammatory infiltrate. Not only was the PIE procedure effective for managing long-term chronic neurogenic bowel by eliminating or diminishing the fecal impaction, there were also no reported cases of bowel injuries caused by the PIE device.
Treatment of fecal impaction with pulsed irrigation enhanced evacuation
Background and Methodology: The goal of this study was to assess whether pulsed irrigation evacuation (PIE) can prevent disimpaction surgery. For patients ranging in age from 13 to 86 with a massive fecal impaction, the PIE procedure was used to break up the fecal impaction.
Results: The PIE treatment was successful in each case. No morbidity arose from the treatment. By the midpoint in our study, because of the success of this treatment, no patient required hospitalization for impaction. The results led the researchers to conclude that “pulsed irrigation enhanced evacuation has been in our experience a simple, quick, and effective treatment for severe fecal impaction.”
Use of pulsed irrigation evacuation in the management of the neuropathic bowel
Goal of Study: This study sought to determine the safety and efficacy of the pulsed irrigation evacuation (PIE) procedure to manage neuropathic bowel for people with spinal cord injury. In the study, 398 PIE procedures were successfully conducted in both in-patient and out-patient settings.
Results: In the outpatient setting, 162 procedures were performed on four outpatients on a regular basis. These patients could not develop an effective bowel routine with the standard digital stimulation, suppositories, or mini enemas. No correctable pathology was found in these patients to explain the failure of the traditional methods. Two of these received their inpatient rehab during the time of the study, and two were referred from the outpatient SCI clinic due to longstanding failed bowel routines. All procedures were effective and tolerated well. In the in-patient setting, 63 procedures done in the SCI population. 20 patients required only one procedure and 11 required procedures on multiple occasions. Two of these patients required four procedures while an inpatient and eventually went on to be long term users of the PIE due to ineffective bowel routines. Of the 63 procedures, 29 (46%) were done for an ineffective bowel routine, 22 (35%) were done for symptomatic impactions, 12 (19%) were done for asymptomatic impactions.
New treatment for rectal impaction in children: an efficacy, comfort, and safety trial of the pulsed-irrigation enhanced-evacuation procedure
Goal of Study: This clinical trial assessed comfort, efficacy, and safety of the pulsed irrigation evacuation (PIE) procedures in children. In the study, the children ranged in age from 4 to 15 years old with chronic constipation, encopresis, and rectal impaction. No sedation or anesthesia was required for any of the patients.
Results: The results showed that in 78% of the procedures, at least 50 percent of the feces was removed from the colon due to the PIE procedure as assessed by abdominal X-ray. Additionally, no water intoxication, hyponatremia, hypokalemia, or hypophosphatemia was found after the procedure. Overall, the study concluded that PIE is safe, effective, and comfortable for children.
Pulsed irrigation enhanced evacuation: New method for treating fecal impaction
Goal of Study: The goal of this study was to determine if pulsed irrigation evacuation (PIE) can assist patients with neurogenic impairment to clear fecal impactions. This study included patients with cerebrovascular accidents, spinal cord injuries, Parkinson’s disease, and head injuries.
Results: No patients reported pain, and patients with diabetes did not become hypoglycemic. This study concluded that the PIE product is safe and effective on an array of patients with neurogenic bowel secondary to spinal cord trauma or neurologic condition. https://pubmed.ncbi.nlm.nih.gov/1929815/
